|
Epoxyeicosatrienoic acids activate transglutaminases in situ and induce cornification of epidermal keratinocytes. 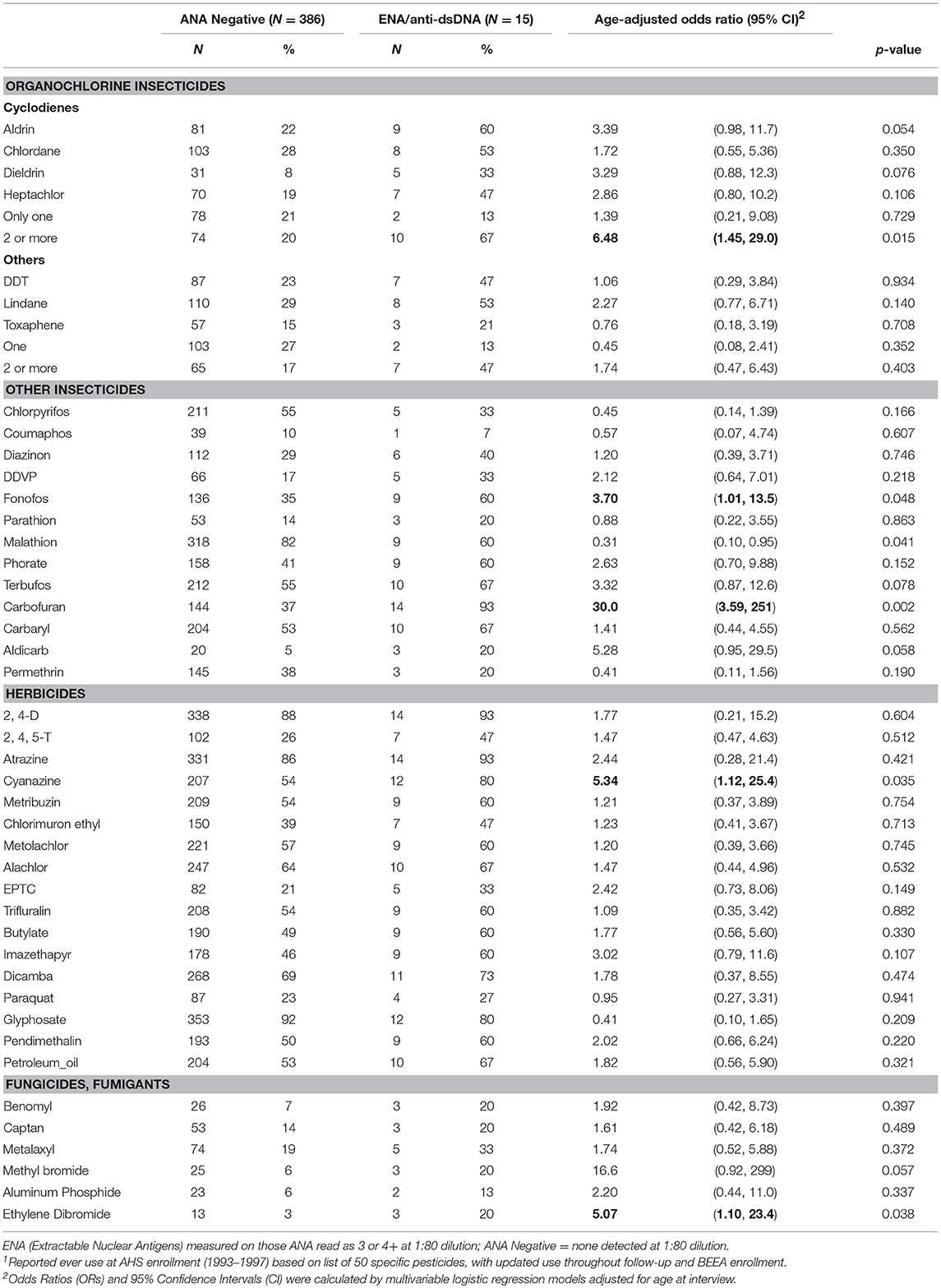
« lessĮpoxyeicosatrienoic acids produced by mouse CYP2B19 have been implicated in mechanisms regulating epidermal cornification (Ladd, P.A., Du, L., Capdevila, J.H., Mernaugh, R., Keeney, D.S., 2003. The lack of catalytic activity of 3A4 toward the t-3,4-dihydrodiol, despite its high activity toward CPP oxidation to tetrahydrotetrols, suggests the possibility of two dihydrodiol epoxides, 3,4-dihydrodiol 9,10-epoxide and 9,10-dihydrodiol 3,4-epoxide, of CPP. The 9,10 K-region double bond was not efficiently oxidized by cytochrome P450 1A1, but by P450 1A2 either from CPP or from the t-3,4-dihydrodiol. It was found that the 3,4-cyclopenta double bond can be oxidized by the cytochrome P450 enzymes 1A1,1A2, and 3A4. In an attempt to explain the more » variation among human liver samples, the microsomes derived from genetically engineered cells containing specific cytochrome P450 isozyme cDNAs were employed. Even though all three human samples were capable of producing all the metabolites identified from the mouse liver microsomal incubations of CPP, the quantity of each metabolite varied among the microsomal samples. Five new metabolites recently identified in our lab, 4-hydroxy-3,4-dihydroCPP,3,4-dihydroCPP-cis-3,4-diol, 4-oxo-3,4-dihydroCPP, 3,4,9,10-tetrahydroCPP-trans-3,4-trans-9,10-tetrol, and trans-3,4-dihydroCPP-3,4,x-triols, as well as the known major metabolite, 3,4-dihydroCPP-trans-3,4-diol, were all observed from the incubations of human liver microsomes and CPP. Human liver microsomes from three individuals were examined, as well as CD-1 mouse liver microsomes. The metabolism of the environmental pollutant and suspected human carcinogen, cyclopentapyrene (CPP), was investigated. Cumulative inhibition of P450 2B6 by mixtures of four low-dosed insecticides.These findings indicate the metabolism of pyridostigmine bromide is unlikely to be under any component of sexual = in the nanomolar range. Using unlabelled and 14C-pyridostigmine, metabolite formation was not observed in both male and female rat and human subcellular fractions, specifically cytosol and S9, or under conditions favoring human FMO activity (pH 8.3). The following CYP-450 substrates were co-incubated with pyridostigmine: phenacetin, tolbutamide, chlorzoxazone, bufuralol, and testosterone. Indirect evidence that pyridostigmine metabolism is not via the major human hepatic CYP-450s involved in drug metabolism, 1A2, 2C9, 2E1, 2D6, and 3A4, was observed by failure to inhibit these isozymes while co-incubated with substrates specific for those isozymes at concentrations of 2-3 times Km. 
Using male and female microsomes from both rat and human, our data suggest that pyridostigmine bromide is not metabolized by the human live microsomes or DNA expressed human CYP-450s via direct observation of no metabolites being formed for incubations up to 90 minutes. A reverse phase High Pressure Liquid Chromatography (HPLC) method was developed to separate pyridostigmine bromide from four potential metabolites.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
